
For complete indications for use, contraindications, warnings, precautions, and side effects, call 866.360.4747 or visit .Ĭaution: U.S. Your doctor may be able to provide additional information on the Boston Scientific Spinal Cord Stimulator systems. subperception therapy) it is less likely that sudden stimulation changes resulting in distraction could occur while having stimulation on when operating moving vehicles, machinery, and equipment. For therapy that does not generate paresthesia (i.e. Stimulation must be turned off first in such cases. Patients using therapy that generates paresthesia should not operate motorized vehicles such as automobiles or potentially dangerous machinery and equipment with the stimulation on. Advise your physician that you have a Spinal Cord Stimulator before going through with other implantable device therapies so that medical decisions can be made and appropriate safety measures taken. The Spinal Cord Stimulator system may interfere with the operation of implanted sensing stimulators such as pacemakers or implanted cardiac defibrillators. The system should not be charged while sleeping. Strong electromagnetic fields, such as power generators or theft detection systems, can potentially turn the stimulator off, or cause uncomfortable jolting stimulation. As a Spinal Cord Stimulation patient, you should not have diathermy as either a treatment for a medical condition or as part of a surgical procedure. Exposure to MRI may result in dislodgement of the stimulator or leads, heating of the stimulator, severe damage to the stimulator electronics and an uncomfortable or jolting sensation. 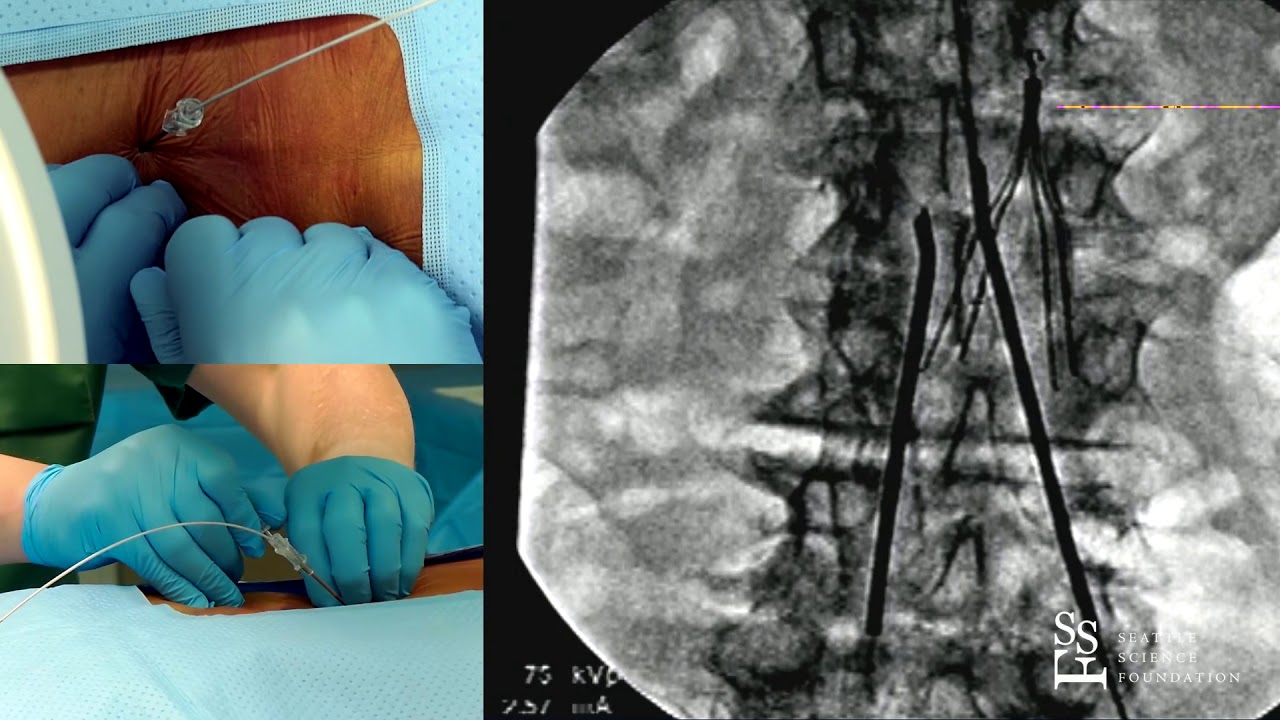 
Patients implanted with Boston Scientific Spinal Cord Stimulator Systems without ImageReady™ MRI Technology should not be exposed to Magnetic Resonance Imaging (MRI). For OTW’s coverage of this expansion see “ Nevro Expands Spine Cord Stim Mfg to Costa Rica.Warnings. Growth does appear to be in Nevro’s future with plans to expand its manufacturing to Costa Rica. Other potential risks include “market related issues, new competition, slower than expected market growth.” Wells Fargo research analysts identified several risks for Nevro including its patent litigation. This is because Nevro has new management and it believes that the spinal cord stimulation market “remains a healthy and underpenetrated market.” Furthermore, Wells Fargo believes that Nevro “has a strong and under-appreciated pipeline.” In its equity research, it indicated that it believes Nevro’s stock should perform better in the future. Wells Fargo research analysts seem to have confidence in Nevro. At present, a hearing is scheduled for January 2021. Summary judgment rulings and other pressures on the parties help courts encourage parties to settle and clear dockets. Additionally, the consultant highlighted that the COVID-19 pandemic has created a backlog that the courts are eager to clear. It was pointed out that many of the issues in the matter have already been addressed by the Federal Circuit. Wells Fargo’s patent consultant suggested that the court might be motivated to decide the case on summary judgment allowing for a speedy resolution. In its research, Wells Fargo stated that its patent consultant believes “that Nevro’s patents are valid.” This optimism and recent court rulings in Nevro’s favor may prove significant in the future. Boston Scientific does not currently provide a high-frequency spinal cord stimulation system in the United States. The litigation revolves around several of Nevro’s patents related to its high-frequency spinal cord stimulation system. and Boston Scientific Corporation indicating the potential for a speedy resolution. October 2020 Wells Fargo Securities, LLC (Wells Fargo) provided an update to the ongoing patent litigation between Nevro Corp.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
